A single-file molecular highway: Mimicking biological channels to extract heavy metals
Imagine an exclusive event where the invited VIPs and the party-crashers look virtually identical, wearing the exact same outfits and standing at the exact same height. How do you instantly let the true VIPs through while blocking the imposters? That is essentially the challenge of separating critical heavy metals like uranium, copper, and gold from complex industrial liquids, where they are mixed with highly similar competing ions.
Today, industries rely heavily on solvent extraction to separate these valuable metals, a process that requires massive amounts of harsh chemicals and takes a significant environmental toll. While membranes offer a cleaner, chemical-free alternative, they have historically failed at this specific task because heavy metal ions are often so similar in size and charge that standard filtration cannot tell them apart.
To solve this, researchers from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT), Chinese Academy of Sciences, looked to a master of microscopic sorting: the biological cell.
The Bouncer and the Highway
Specifically, the team learned from biological voltage-gated calcium (CaV) channels. These channels act like an exclusive VIP club. They feature a narrow, one-dimensional hallway lined with highly specific binding sites (Fig. 1a). When the "VIP" calcium ions enter in a single file, they effectively block the door for all other competing ions—a phenomenon scientists call the "anomalous mole fraction effect" (AMFE).
"If these ions are bound so tightly to the channel walls, how do they move through it so incredibly fast?" explains Jun Gao, corresponding author of the study and researcher at QIBEBT. "The answer is microscopic shoving. Because the ions are crammed into a tiny, single-file space, they strongly repel each other, essentially pushing the ion in front of them rapidly toward the exit."
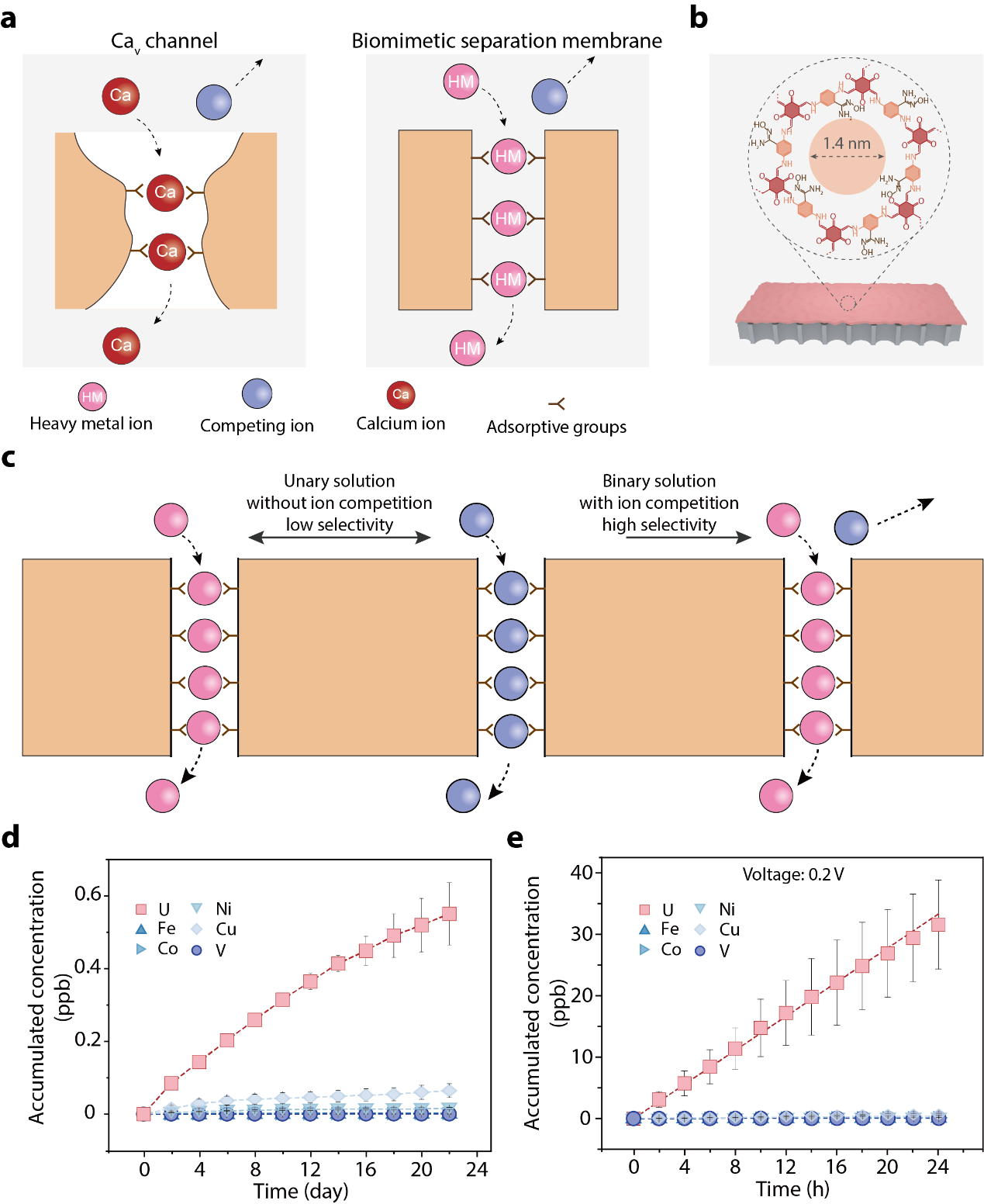
Fig. 1 Bioinspired membrane separation of heavy metal ions.a, Schematic illustration of ion transport behavior of biological calcium channel and bioinspired membrane. b, Molecular structure of COF membrane. c, AMFE of the artificial ion channels. d, Accumulated concentration of uranium diffused from natural seawater over 22 days. e, Accumulated concentration of uranium from seawater electrodialysis.
Building an Artificial VIP Lane
Inspired by this elegant natural design, the research team engineered a new separation mechanism using covalent organic frameworks (COFs).
"We created microscopic channels just wide enough, at about 1.4 nanometers, to force target heavy metal ions to line up in a single file," notes Yongye Zhao, co-first author of the paper. Hongfei Gao, also a co-first author, adds that this precise confinement mitigates the standard size exclusion effect while perfectly accommodating the required single-file transport.
When the researchers coated the insides of these artificial channels with specific chemical "bait"—amidoxime groups designed to attract uranium—the results were striking (Fig. 1b). The system successfully mimicked the biological AMFE (Fig. 1c): once uranium entered the channel, it blocked out competing elements like vanadium. And because the trapped uranium ions repelled one another, they shuttled through the barrier smoothly and rapidly, bypassing the usual gridlock.
From Ocean Water to Gold Mines
The real-world potential of this mechanism is vast. In a continuous test using natural seawater over 22 days, the process efficiently pulled out uranium while rejecting a sea of other background metals (Fig. 1d). By using a low-voltage electrodialysis method (Fig. 1e), the strategy achieved an extraction throughput at least an order of magnitude higher than traditional adsorbents.
Furthermore, the team proved this bioinspired strategy is highly versatile. By changing the chemical functional groups inside the channels, they successfully extracted copper—a crucial component for clean energy technologies—directly from highly acidic simulated mining leachates. They also adapted the chemistry using bipyridine to selectively filter and separate gold from complex liquid mixtures.
"By turning passive adsorptive materials into active, single-file highways for specific metals, this approach bridges the gap between traditional chemical adsorption and modern water filtration," says Chaoxu Li, another corresponding author of the study.
Ultimately, this microscopic sorting mechanism could one day lead to a much greener, more sustainable global mining and recycling industry.
The study published in the journal Nature Nanotechnology. Dr. Yongye Zhao and Dr. Hongfei Gao from QIBEBT are the co-first authors of the paper, with Prof. Jun Gao and Prof. Chaoxu Li serving as corresponding authors. The work was supported by the National Natural Science Foundation of China and the QIBEBT/SEI/QNESL.
(Text/Image by GAO Jun ZHAO Yongye)