Revealing an Unheeded Failure Mechanism of Mg Metal Anode
Among various post-lithium-ion battery technologies, rechargeable magnesium (Mg) batteries utilizing divalent Mg2+ as charge carriers are expected to offer substantial improvements in volumetric energy density and sustainability of batteries.
Now, a research team led by researchers from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT), Chinese Academy of Sciences (CAS), has revealed an unheeded failure mechanism of Mg metal anode, which may promote the development of Mg metal anode battery from a new perspective.
This study was published on June 8 in Advanced Materials.
"The initial plating Mg can be undoubtedly uniform in wide range of current densities and under a practical areal capacity. However, an unusual self-accelerating pit growth is observed in the Mg stripping side, which severely deteriorates the anode integrity and subsequent Mg plating uniformity, causing Mg metal anode failure or short circuit of battery," said LIU Xin, doctoral candidate at QIBEBT and lead author of the study.
The research team utilized macroscopic and microscopic characterization methods to investigate the plating/stripping behavior of Mg metal anode under various current density. The experimental results proved the dendrite-free advantage of Mg metal anode in a classic electrolyte. Unfortunately, the team proposed that uneven stripping behavior was inevitable in the chlorine-containing electrolytes, though the chlorine seemed to be beneficial for the plating process.
The team pointed out that the degree of uneven stripping versus the current density displayed a volcano plot. Extremely low or high current density would generate relatively uniform stripping morphology. Furthermore, the team proved that stirring electrolyte and adding leveling agent would improve the stripping behavior.
"The study of Mg metal anode stripping behavior illuminated a non-dendrite-induced Mg battery failure mechanism and provided effective strategies to solve this puzzle," said Prof. CUI Guanglei from QIBEBT.
The study was supported by the National Natural Science Foundation of China, Natural Science Foundation of Shandong Province, Strategic Priority Research Program of Chinese Academy of Sciences, China Postdoctoral Science Foundation, Taishan Scholars Program for Young Expert of Shandong Province and Shandong Energy Institute.
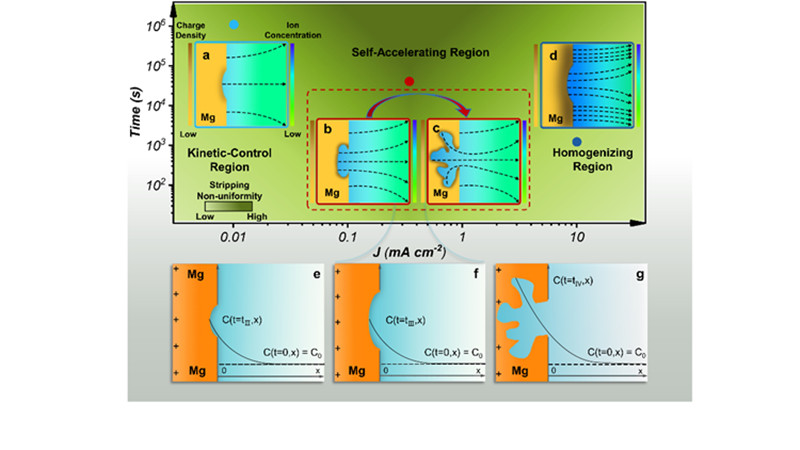
The stripping morphologies and distributions of chlorine-containing ions at the Mg metal anode-electrolyte interface at different current density. (Image by LIU Xin)
(Text by LIU Xin)
Contact:
CHENG Jing
Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences
Tel: 86-532-80662647/80662622
E-mail: chengjing@qibebt.ac.cn