Metagenomics Used in Biological Ingredient Analysis of Traditional Chinese Medicine Preparation
Traditional Chinese Medicines (TCMs) have long been widely utilized to prevent and treat various diseases in China and many other countries. A “TCM preparation” (or “patented drug”) is characterized by the utilization of multi-herbal materials (including medicinal plants, animal materials and mineral) with their respective dosages by the guidance of Chinese medicine theory. Generally the TCM ingredients assessment methods include biological component (i.e., species composition) analysis and chemical analysis. However, current analysis of the TCM ingredients mainly focuses on chemical methods based on spectroscopy and chromatography, which cannot identify the contaminated ingredients.
The joint research effort from Single Cell Center of Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences (QIBEBT-CAS) and Shandong Academy of Medical Sciences has developed a method for biological assessment of the quality of TCM preparations based on high-throughput sequencing and metagenomic analysis, and also tested this method on Liuwei Dihuang Wan (LDW, 六味地黄丸) for both prescribed species and contaminated species. Results have been published online in Nature Publishing Group's Scientific Reports (Cheng, Su, et al, Scientific Reports, 2014) on June 3, 2014.
Assistant Professor SU Xiaoquan and Master student CHENG Xinwei have proposed the method (M-TCM) for comprehensive analysis of prescribed species and contaminated species of the TCM sequencing data using ITS2 and trnL biomarkers based on high-throughput sequencing and metagenomic analysis methods. By this method, different batches of Liuwei Dihuang Wan from different manufacturers have been compared based on the quality assessment of their biological ingredients, and their qualities have been evaluated.
Since M-TCM is a general approach, it can identify species based on molecular markers and bioinformatics analysis to accurately detect the contaminated species in TCM preparations without professional background. Therefore, it has a great application potential in bio-medicine and related components analysis. Researchers will continue in the integration of biological and chemical composition analysis for more comprehensive assessment of the quality of TCM.
The above research works have been published in Scientific Reports, Genomics, Proteomics & Bioinformatics, and the patent applications are also in process. The works were led by Professor NING Kang and Associate Professor BAI Hong (Shandong Academy of Medical Sciences).
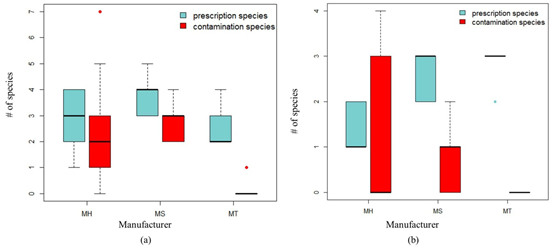
Number of prescribed species and contaminated species detected in LDW samples from different manufacturers. (a) Species composition analysis based on ITS2 sequencing results. (b) Species composition analysis based ontrnLsequencing results.(Image by Single Cell Center, QIBEBT)
Reference:
(1) Biological ingredient analysis of traditional Chinese medicine preparation based on high-throughput sequencing: the story for Liuwei Dihuang Wan,Scientific Reports, 4:5147, doi:10.1038/srep05147.
Contact:
Prof. NING Kang
Email: ningkang (AT) qibebt.ac.cn